 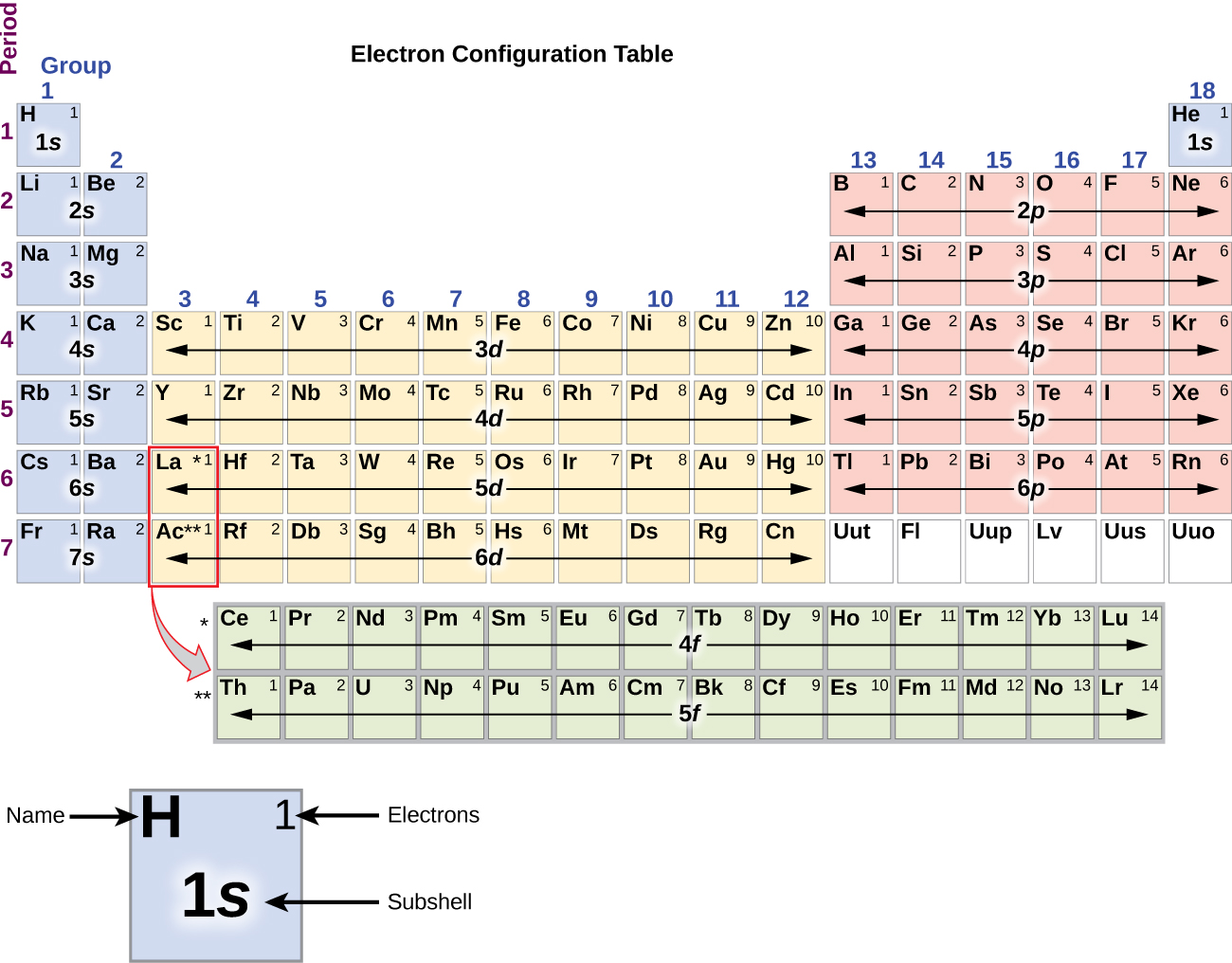  
Unfortunately, there was a slightly different system in place in Europe. The first two groups are 1A and 2A, while the last six groups are 3A through 8A. How to determine the number of valence electrons and draw Lewis structures for main group elements starting from the electron configuration. The traditional system used in the United States involves the use of the letters A and B. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. In groups 112, the group number matches the number of valence electrons in groups 1318, the units digit of the group number matches the number of valence electrons. These dots are arranged to the right and left and above and below the. The number of dots equals the number of valence electrons in the atom. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. A Lewis electron dot diagram (or electron dot diagram, or a Lewis diagram, or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. Naturally occurring potassium is composed of three isotopes, of which 40K is. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. All of the alkali metals have a single valence electron in the outer electron shell, which is easily removed to create an ion with a positive charge a cation, which combines with anions to form salts. A new period begins when a new principal energy level begins filling with electrons. In the periodic table, potassium is one of the alkali metals. There are seven periods in the periodic table, with each one beginning at the far left. \) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
